MEMBER NEWS
Is NICE decision-making for rare diseases failing patients?
July 16th, 2021
The NICE Highly Specialized Technologies (HST) appraisal is the gatekeeper for whether innovative treatments for ultra-rare diseases are approved in England. But does this process support patient access to new drugs as effectively and fairly as it should? In this article, we weigh the balance of how well the NICE submission process is working for payers, providers, patients and those that care for them.
Since 2019, Pfizer’s Vyndaqel drug has been achieving extraordinary results in US for a rare heart condition, ATTR-CM. As reported in the trade press in May 2020, Vyndaqel has been shown to reduce death and hospitalizations in patients with transthyretin amyloidosis with cardiomyopathy (ATTR-CM). It is the only drug in the world to treat the disease, which often goes underdiagnosed because its symptoms are so subtle.
Approximately 800 people have been diagnosed with ATTR-CM in England, with the manufacturer proposing a list £130,000 annual cost, but with a substantial (albeit confidential) discount.
NICE made its original decision to reject the drug in October 2020 and rejected Pfizer’s appeal last month. Both NICE and Pfizer say they are still open to negotiation. Patient advocacy groups Cardiomyopathy UK and the British Society for Heart Failure both supported Pfizer’s unsuccessful appeal against the original NICE decision.
Sadly for patients, this process of brinkmanship between drug manufacturer and NICE is all too familiar. The graphic below shows the timeline for the first 12 drugs assessed under the HST system, of which 10 were eventually approved. The average time from submission to published assessment was 17.7 months. This is slowly improving, as submitting companies and NICE streamline their efficiency. (Source: Simon-Kucher & Partners, NICE).
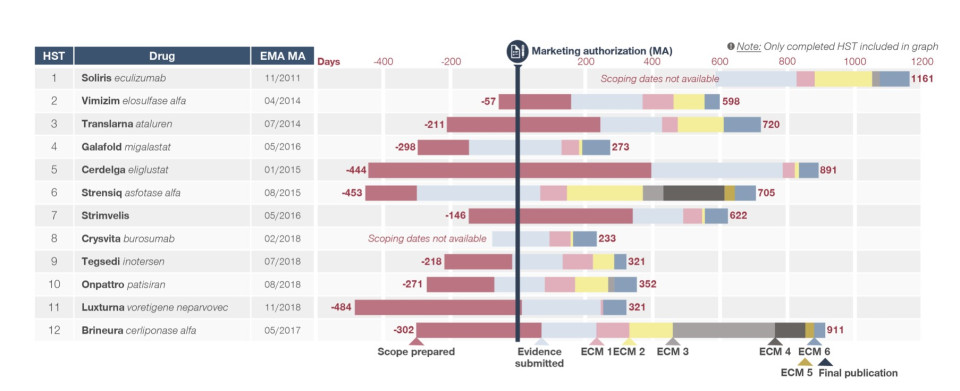
Yet, as recently as 2019, the cause celebre example is Vertex’s Orkambi cystic fibrosis drug, which took four years to resolve between NHS England, Vertex and NICE, due in part over whether the STA or HST model would be the most appropriate for assessment, as well as a row over the NICE requirement for full assessment of data.
HST Approval timeline
Vertex asked a premium price of over £100,000 per year whilst NHS England offered just 10 percent of that. NICE used its Single Technology Appraisal (STA) system for assessment, which is used for all but the ultra-rare conditions. Non ultra-rare diseases find the STA system very challenging, and there is a strong argument for a middle-tier appraisal system. Cardiomyopathy UK research in 2019 has shown that only 9% of people living with cardiomyopathy are receiving the ideal care and treatment.
It is hard to deny the vital role that NICE plays in trying to maximise the value that NHS can achieve for patients in England, given the limited total NHS budget available. That said, it's ‘two-size-fits-all’ system typically supports ultra-rare disease treatments under the highly specialised technology (HST) appraisal that achieve quality-adjusted-life-years (QALY) for annual costs below a £300,000 ceiling – a test that Vyndaqel would likely have passed. Orphan rare diseases now account for 16% of global prescription sales, highlighting that it is both a significantly high cost, but also that perceiving rare diseases as only affecting a few people is misleading – c.300m people live with a rare disease (around 4 per cent of the world’s population).
To be fair to NICE, its appraisal systems are by no means ‘set in stone’, and have been amended over the years, such as the shift from considering life extending treatments used at the end of life towards the “burden of illness” which reflects the loss (or shortfall) in quality and length of life, as a consequence of having a disease or condition. It also has incorporated the ‘wider societal impact’ of a new treatment, to impact a person’s capacity to engage with society.
Another significant change has been to recognise and include carer health-related quality of life in its technology and HST appraisals. For many rare diseases, especially for child patients, the impact of carer quality of life can be very significant.
However, in a 2019 analysis of how many NICE submissions actually included data to support carer quality of life, only 16 of 422 appraisals has such information provided. Part of the reason for this is the difficulty in gathering data across the limited evidence base associated with rare disease populations. As such, submitting pharma companies often have to rely on clinical expert opinion to address the evidence gaps – these clinicians typically do not have any caregiver data to help support a NICE submission case.
This finding highlights how NICE decision-making is a ‘two-way-street’ – whilst it can make its appraisal system more inclusive and context-sensitive, it can only make its decisions based on the data provided to it.
So - is NICE decision-making for rare diseases really failing patients?
1) NICE’s two-tier appraisal system is helpful for ultra-rare conditions, but makes many non-ultra-rare disease treatments very challenging to win approval for, when assessed under the STA system. When such submissions fail, pharma companies and NICE can often end up in a protracted battle of contract and price-haggling, leaving patients waiting for drugs that have proven quality-of-life improvement.
2) NICE is responsive to considering changes to its formula for assessment calculations, and a ‘third-way’ built for non-ultra-rare disease treatments could be a valuable and fair way to improve patient access, and in so doing encourage more investment from pharma companies in rare diseases.
3) Within the existing NICE appraisals, it is critical for patients that submitting pharmaceutical companies include research data to support the assessment of treatments on carer quality of life, as well as the patient. Pharma companies could do more to develop such carer impact data, including working with research consultancies and patient advocacy groups to capture and aggregate quality-of-life experiential data.
Rick Harris
Customer Faithful Ltd
References:
1) https://www.fiercepharma.com/pharma/a-costly-decision-nice-rejects-pfizer-heart-disease-drug-vyndaqel-but-open-to-negotiation
2) http://nicedsu.org.uk/wp-content/uploads/2019/07/2019-04-03-NICE-carer-HRQL-v-2-0-clean.pdf
Disclaimer: The views and opinions expressed in this feature are those of the author and may not reflect the official policy or position of the BHBIA. The BHBIA have not verified any of the information quoted and do not accept any responsibility for its accuracy, or otherwise.